 
Investigators therefore concluded that pembrolizumab is an effective first-line therapy in patients with metastatic or locally advanced NSCLC with greater than 1% PD-L1 TPS who do not have other sensitizing mutations or translocations. Similar treatment-related death rates occurred. Treatment-related adverse events that were grade 3 or higher were significantly less common in the pembrolizumab group (18%), as compared to the chemotherapy group (41%). Researchers found that pembrolizumab prolonged overall survival in all three analyses, as compared to chemotherapy, yielding a hazard ratio for death of 0.69 in those with 50% or greater PD-L1 TPS (95% CI 0.56 to 0.85, p=0.0003), 0.77 in those with 20% or greater PD-L1 TPS (95% CI 0.64 to 0.92, p=0.0020), and 0.81 in those with 1% or greater PD-L1 TPS (95% CI 0.71 to 0.93, p=0.0018). Analysis was performed on three separate groups, those with PD-L1 TPS of 50% or greater, 20% or greater, and 1% or greater. 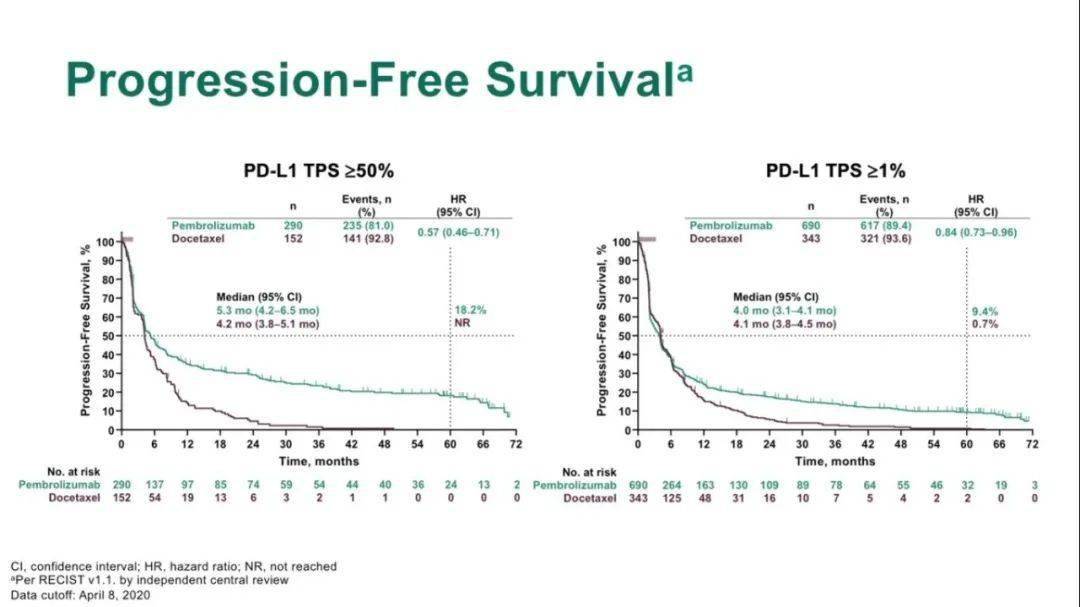 
They were randomized to receive either pembrolizumab (200 mg every 3 weeks for up to 35 cycles) or the investigator’s choice of platinum-based chemotherapy (4-6 cycles). Patients were included in the study if they had previously untreated locally advanced or metastatic NSCLC, expressed PD-L1 as above, had an Eastern Cooperative Oncology Group (ECOG) performance score of 0 or 1, and did not have a sensitizing EGFR mutation or ALK translocation (n=1,274). In this randomized, open-label, phase 3 study, pembrolizumab was investigated for its efficacy in prolonging survival in locally advanced or metastatic NSCLC with a PD-L1 TPS of 1% or greater. Previous studies have shown that pembrolizumab is effective in prolonging survival in advanced non-small-cell lung cancer (NSCLC) with a PD1-ligand (PD-L1) tumor proportion score (TPS) of 50% or greater. As a matter of fact, this is has just been published by in The Lancet just about last week.Pembrolizumab is a monoclonal antibody against programmed cell death protein 1 (PD-1). So that pretty well gives you a capsule of what we have learned from this important randomised study. This group of patients then I think we can consider the single agent use as per 042. On the other hand, there are some patients who are physically not capable of receiving chemotherapy. So this group probably can be considered the utilisation of a KEYNOTE-189 regimen. So I wouldn’t promote to replace chemotherapy with the single agent pembrolizumab and especially there are data from KEYNOTE-189 demonstrating that the combination of chemo with pembrolizumab is actually superior to chemotherapy. Now, we come to the question that can we use single agent pembrolizumab in the 1-49% subgroup as a first line? In this study we actually did not show any superiority but then there is some advantage in terms of toxicity. In KEYNOTE-042, pembrolizumab monotherapy likely had a worse PFS than chemotherapy for patients with PD-L1 of 149. So, in other words, whether you use 1% or 20% the benefit is mostly driven by the 50% subgroup. With this one we did not observe any overall survival benefit. But then, speaking of that, we also performed an exploratory analysis looking at the subgroup of 1-49%. At the same time we also find that there is an improvement in the over 20% cut-off and the over 1% cut-off. Now, the 50% subgroup of patients the advantage is actually similar with a very nice hazard ratio although the median is not as impressive as what we observed in 024. We were actually able to demonstrate an overall survival benefit when we looked at using a cut-off at 1%, 20% or 50%. In this randomised study we actually compared patients with over 1% expression, single agent pembrolizumab versus standard chemotherapy. Now, the importance of KEYNOTE-042 is, number one, it will support the efficacy of the patient over 50% and, second, it’s to say how about if we use 1% as a cut-off, are we going to observe an overall survival benefit? So those are the major differences about these two studies. KEYNOTE-042 is a similar design however we select the patients over 1% and using overall survival as the primary endpoint. So KEYNOTE-024 is actually selecting the patients of PD-L1 expression 50% or more and a primary endpoint of progression free survival. 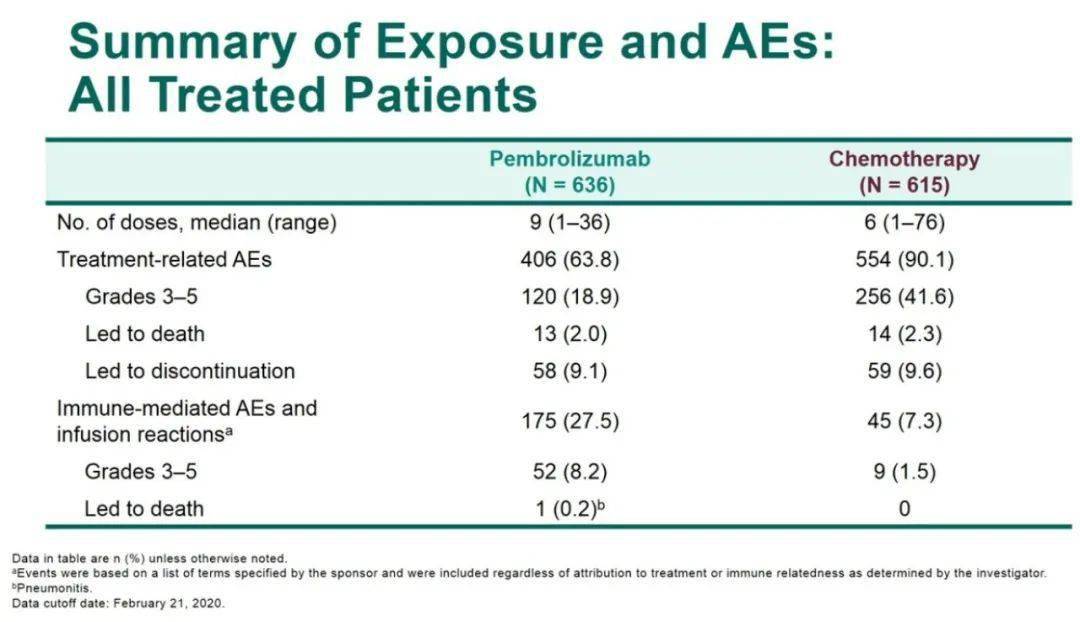
KEYNOTE-042 is the sister trial of KEYNOTE-024 – 24, 42. Pts received pembro 200 mg Q3W for 35 cycles or chemo Q3W for 4-6 cycles (optional pemetrexed maintenance for nonsquamous). Professor Tony Mok – Chinese University of Hong Kong, Hong Kong Eligible Asian pts were enrolled in China, Hong Kong, Japan, South Korea, Malaysia, Philippines, Taiwan, Vietnam and Thailand in the KEYNOTE-042 global (NCT02220894) and China extension (NCT03850444) studies. Latest data from KEYNOTE 42: Pembrolizumab vs chemotherapy in treatment-naive patients (pts) with PD-L1 advanced NSCLC 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
